Alkanes
Alkanes are Saturated Hydrocarbons. They consist of the elements Carbon and Hydrogen, and all the bonds between the Carbon atoms are single bonds. They have the general formula CnH2n+2. Since all Alkanes are similar in structure and properties, they are called a Homologous Series.
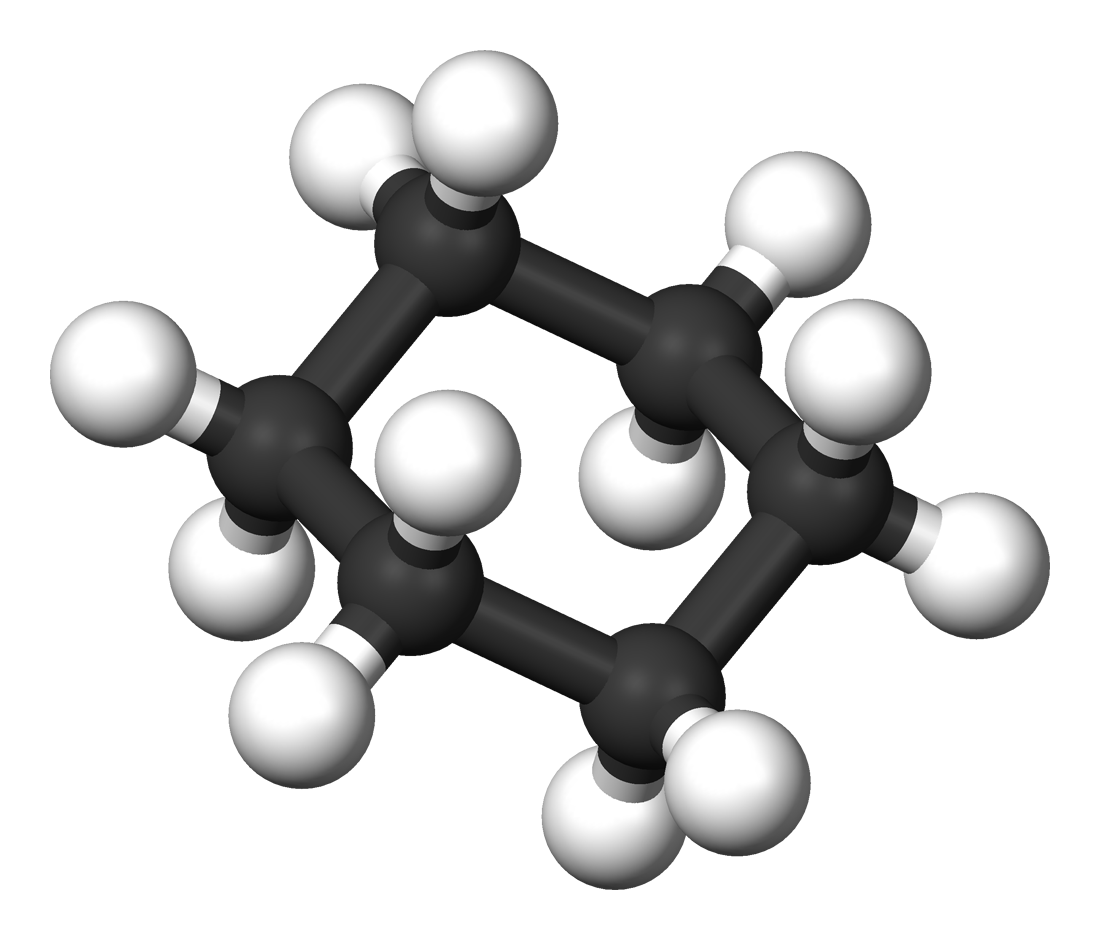
The structure of an Alkane, as with all Hydrocarbons, can be represented with a
- A 3D model, with all the atoms and bonds shown, modelled in 3 dimensions.
- Full Structural Formula, with all the atoms and bonds shown.
- Skeletal Formula, with only the bonds between the Carbon atoms shown.
- Shortened Structural Formula, a linear form where all the atoms are shown but no bonds.
Branched Alkanes and Cycloalkanes
Alkanes can be long straight chains of Carbon atoms bonded with each other and Hydrogen, but they can also come in different shapes. Branching can occur, as can the formation of cyclic structures. This is called Isomerism.
Branched Alkanes occur when a straight chain Alkane forms replaces one of its Hydrogen atoms with an Alkyl group (an Alkane which has similarly lost a Hydrogen on the end Carbon), forming a branch. Branching can happen in many different ways and means that a variety of possible structures can exist for one Molecular Formula. Branched Alkanes may have the prefix ‘iso’.

- Cyclic Alkanes have the general formula CnH2n and consist of Carbon atoms bonded in a circular structure. They are name with the prefix ‘cyclo’, for example cyclohexane.
Reactions of Alkanes
Since Alkanes are Saturated Hydrocarbons, they do not readily react. However, they can be made to react under certain conditions, so give useful products or energy output.
Alkanes will react with Oxygen if they are given sufficient Activation Energy. This will result in a highly Exothermic reaction, producing Carbon Dioxide and Water, which makes Alkanes very useful as fuels.
When Alkanes C4-C6 are heated to 150°C with a Platinum of Aluminium Oxide Catalyst, Isomerism occurs. The reaction produces Branched Alkanes, and so is useful in improving Octane Number.
Reforming is the process of increasing the amount of Cycloalkanes and Hydrocarbons containing Benzene Rings, to improve Octane Number. This is done with Alkanes in the Naphtha Fraction (C6-C10) at 500°C with a Platinum or Aluminium Oxide Catalyst. Hydrogen is recycled through the mixture to reduce ‘coking’.
Cracking is the process of breaking longer chained Alkanes down into smaller Alkanes and Alkenes, sometimes for Polymer manufacture, and sometimes to improve Octane Number.
Steak Cracking involves heating Alkanes from the Naphtha and Kerosene Fractions (C6-C16) to 900°C without a catalyst and using Steam as a diluent to reduce ‘coking’. This is used in the manufacture of Polymers.
Catalytic Cracking takes feedstock from longer chained Alkanes in the Gas Oil Fraction (C14-C20) and heating to 500°C with a Zeolite. This can produce Branched and Cyclic Hydrocarbons and is used to improve Octane Number.