Lipids
Lipids are made of the elements Carbon , Hydrogen and Oxygen, although they have a much lower proportion of water than other molecules such as Carbohydrates. They are insoluable in water
Lipids perform many functions, such as:
- Energy Storage
- Making Biological Membranes
- Insulation
- Protection - e.g. protecting plant leaves from drying up
- Boyancy
- Acting as hormones
They are made from two molecules: Glycerol and Fatty Acids.
A Glycerol molecule is made up from three Carbon atoms with a Hydroxyl Group attached to it and Hydrogen atoms occupying the remaining positions.

Fatty Acids
- Fatty acids consist of an Acid Group at one end of the molecule and a Hydrocarbon Chain, which is usually denoted by the letter ‘R’.

- Fatty acids may be saturated or unsaturated. A fatty acid is saturated if every possible bond is made with a Hydrogen atom, such that there exist no C=C bonds. Saturated fatty acids on the other hand do contain C=C bonds. Obviously monounsaturated fatty acids have one C=C bond, and polyunsaturated have more than one C=C bond.
If fatty acids are unsaturated, their shape is altered from a saturated molecule so the molecules in the Lipid push apart, thus making it more fluid and oily.
Animals tend to have more saturated, and consequently solid at room temperature lipids whereas plants tend to have more unsaturated and so fluid at room temperature lipids.
Triglycerides
- Triglycerides are lipids consisting of one glycerol molecule bonded with three fatty acid molecules. The bonds between the molecules are covalent and are called Ester bonds. They are formed during a condensation reaction.
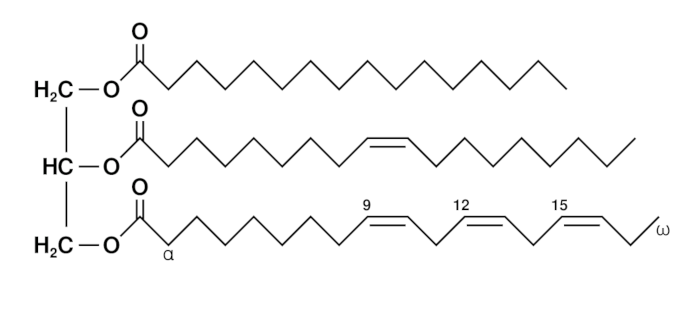
- Triglycerides are hydrophobic and so insoluble in water. The charges are evenly distributed around the molecule so hydrogen bonds to not form with water molecules.
Phospholipids
- Phospholipids are similar to triglycerides in they consist of a glycerol ‘backbone’ and fatty acid ‘tails’, however, the third fatty acid has been replaced by a phosphate group ‘head’.

While the fatty acid ‘tails’ are hydrophobic, the phosphate ‘head’ is hydrophilic. This means the phosphate group will orientate itself towards water and away from the rest of the molecule, and also gives rise to the special properties that allow phospholipids to be used to form membranes.
Phospholipids can contain saturated and unsaturated fatty acids. This allows for the control of the fluidity of membranes, which is useful, for example, in maintaining membrane fluidity at low temperatures.